The medical industry demands perfection. One tiny flaw in an implant can lead to severe patient complications or massive recalls. Engineers struggle to balance intricate designs with strict biocompatibility requirements. This high-stakes environment makes choosing the right manufacturing partner stressful and complex.
CNC machining for medical implants requires adherence to rigorous standards like ISO 13485 and ASTM F136. Manufacturers must use certified biocompatible materials such as Titanium Grade 23 (Ti-6Al-4V ELI) or PEEK. Success depends on maintaining strict traceability, achieving surface finishes below 0.8 Ra, and ensuring zero contamination throughout the production process.

Choosing the right material is just the first step. You also need to navigate a maze of regulatory standards. If you miss one documentation requirement, your entire project could stall. Let’s break down exactly what you need to know to get your medical components made correctly and legally.
Which Materials Are Safe and Effective for Long-Term Implantation?
Selecting the wrong material for an implant is not just an engineering failure; it is a safety hazard. Corrosion, rejection by the body, or mechanical fatigue can cause catastrophic failure. Engineers must find materials that are strong, lightweight, and completely biocompatible.
The best materials for long-term implants are typically Titanium Grade 23 (Ti-6Al-4V ELI) for bone integration and PEEK for spinal cages. Cobalt Chrome is excellent for high-wear joints like knees. Stainless Steel 316LVM is used for temporary fixation but is less common for permanent implants due to weight and stiffness issues.

Material selection goes far beyond just checking a datasheet. It requires understanding how the material interacts with human tissue and the machining process itself.
The Titanium Advantage vs. PEEK
Titanium is the king of medical metals. Specifically, Grade 23 (ELI – Extra Low Interstitials) is preferred because it has fewer impurities like oxygen and iron. This makes it less brittle and more resistant to cracking than standard Grade 5. I remember a project where a client wanted to save money by using standard Grade 5 for a dental abutment. We had to explain that the risk of micro-fractures in the mouth environment was too high. They switched to Grade 23.
However, metal isn’t always the answer. PEEK (Polyether ether ketone) is a high-performance plastic that mimics the stiffness of human bone better than titanium. This prevents "stress shielding," where the bone weakens because the implant takes too much load.
Here is a quick comparison of common implant materials:
| Material | Best Application | Key Benefit | Machining Difficulty |
|---|---|---|---|
| Titanium Gr 23 | Bone screws, plates, dental | Biocompatible, osseointegration | High (Heat generation) |
| PEEK | Spinal cages, cranial plates | Radiolucent (invisible on X-ray) | Medium (Stability issues) |
| Cobalt Chrome | Knee/Hip joints | Extreme wear resistance | Very High (Hardness) |
| 316LVM Steel | Trauma plates (temporary) | Cost-effective, ductile | Medium |
When machining these, we must use dedicated coolants. You cannot use the same cutting fluid for medical PEEK that you used for an aluminum car part. Cross-contamination is a deal-breaker.
Why is ISO 13485 Certification Essential for Your Manufacturer?
Working with a shop that doesn’t understand medical regulations is a recipe for disaster. If your supplier cannot prove their process control, your product will never pass FDA or CE approval. You need a partner who speaks the language of quality assurance.
ISO 13485 is the specific quality management standard for the medical device industry. Unlike general ISO 9001, it focuses on risk management and maintaining the effectiveness of processes rather than just customer satisfaction. It guarantees that every step of production is documented, traceable, and repeatable.

Many engineers assume that if a shop is ISO 9001 certified, they can handle medical parts. This is a dangerous assumption.
The Difference Between 9001 and 13485
ISO 9001 is about continuous improvement. ISO 13485 is about consistency and risk. In the medical field, "improving" a process without validating it first can be illegal. If we change a cutting tool path for a bone screw, we have to prove that this change doesn’t affect the surface integrity or heat treatment of the part.
We once had a German client, much like yourself, who needed a complex spinal hook. He asked for the "First Article Inspection" (FAI). But under ISO 13485, FAI is just one part of the validation. We also had to provide:
- IQ (Installation Qualification): Showing our machines are installed correctly.
- OQ (Operational Qualification): Showing the machines operate within limits.
- PQ (Performance Qualification): Showing the process consistently produces good parts.
Process validation is critical. It proves that we can make 1,000 parts exactly the same as the first one.
Traceability is Key
If a patient has a reaction to an implant five years from now, you need to know exactly which batch of titanium that screw came from. We maintain "lot traceability." Every block of material has a heat number. That number follows the part through milling, deburring, passivation, and packaging.
| Document Type | Purpose | Why You Need It |
|---|---|---|
| Material Certs | Proves chemical composition | Confirm ASTM F136 compliance |
| Travelers | Tracks each production step | Pinpoints where errors occurred |
| CMM Reports | Dimensional verification | Proves geometric accuracy |
| Passivation Cert | Confirms surface cleaning | Ensures corrosion resistance |
How Do Surface Finish and Cleanliness Affect Implant Success?
A rough surface on a medical implant can encourage bacteria growth or cause inflammation. Conversely, some implants need a specific roughness to help bone grow onto them. Getting this surface texture exactly right is difficult and requires specialized post-processing.
Surface finish directly impacts osseointegration (bone growth) and infection risk. Generally, mating surfaces need to be extremely smooth (Ra 0.05µm) to reduce friction, while structural surfaces might be roughened. Cleanliness is non-negotiable; parts must be passivated to remove free iron and ultrasonically cleaned.
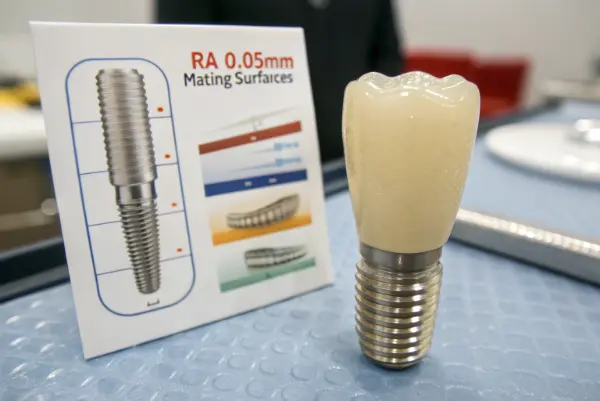
Achieving the right Ra (Roughness Average) value is not just about aesthetics. It is a functional requirement for the human body.
Passivation and Cleaning
After machining, stainless steel and titanium parts often have tiny particles of iron from the cutting tools embedded in the surface. If you implant this, the iron will rust inside the body. This is why Passivation is mandatory. We dip the parts in an acid solution (usually citric or nitric acid). This removes the free iron and builds up a protective oxide layer.
I recently visited a partner shop that claimed to do medical cleaning. I saw them drying parts with a dirty shop rag. That is an immediate fail. Medical parts must be dried in clean environments, often cleanrooms, and handled with gloves to prevent skin oils from contaminating the surface.
Bead Blasting vs. Polishing
Different parts need different treatments:
- Polishing: For ball joints (like a hip replacement ball). We need a mirror finish to prevent wear on the plastic liner. This is often done by hand or automated drag finishing.
- Bead Blasting: For the stem of a hip implant. We blast it with glass beads or hydroxyapatite. This creates a rougher texture. Why? Because bone cells love rough surfaces. They grab onto the pits and hold the implant tight.
Geometric Tolerances
It is not just about the surface feel. The fit must be perfect.
- Tapers: Many modular implants use a "Morse Taper" to lock parts together without screws. If the angle is off by 0.01 degrees, the cold weld won’t happen, and the joint will wobble.
- Threads: Bone screw threads are complex. They often have deep profiles to cut into bone. Machining these in titanium without burrs is an art form. We use thread whirling attachments on our Swiss lathes to get this right.
Conclusion
Manufacturing medical implants requires verified materials like Grade 23 Titanium, strict adherence to ISO 13485 standards, and flawless surface finishing to ensure patient safety and regulatory compliance.
